FAQ's
Frequently Asked Questions
WHAT IS FIRE?
WHAT ARE THE CLASSES OF FIRE?
WHAT IS SMOKE?
WHAT IS IN WOOD?
WHAT TYPE OF FIRE EXTINGUISHER DO I NEED?
WHAT SIZE FIRE EXTINGUISHER DO I NEED?
HOW DO I USE A FIRE EXTINGUISHER?
WHEN CAN'T I USE A FIRE EXTINGUISHER?
HOW DO DRY CHEMICAL EXTINGUISHERS WORK?
WHAT ARE THE THREE A's?
WHAT ARE PHOTOELECTRIC SMOKE DETECTORS?
WHAT ARE IONIZATION SMOKE DETECTORS?
What is Fire?
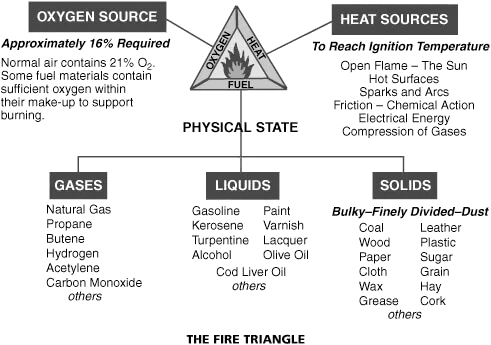
In order to have a fire, there must be three elements:
- Fuel: something which will burn
- Heat: enough to make the fuel burn
- Oxygen: significant levels contained in air
All three elements must be present at the same time to have a fire. Fire will burn until one or more of the elements is removed, then will go out.
Classes of Fires
There are four classes of fires. All fire extinguishers are labeled, using standard symbols, for the classes of fires on which they can be used. A red slash through any of the symbols tells you the extinguisher cannot be used on that class of fire. A missing symbol tells you only that the extinguisher has not been tested for a given class of fire, but may be used if an extinguisher labeled for that class of fire is not available.
CLASS A - fires involve common combustibles such as wood, paper, cloth, rubber, trash and plastics.
CLASS B - fires involve flammable liquids, solvents, oil, gasoline, paints, lacquers and other oil-based products.
CLASS C - fires involve energized electrical equipment such as wiring, controls, motors, machinery or appliances.
CLASS D - fires involve combustible metals such as magnesium, lithium and titanium.
CLASS K - fires involve combustible cooking media such as oils adn grease commonly found in commercial kitchens.
Why does smoke come from a fire?
Let's say you have a nice fire going, and it has burned down to the point where what you see is a collection of hot "glowing embers." The fire is still producing a lot of heat, but it is producing no smoke at all. You might have gotten to this point either by starting with logs in a fireplace or by starting with charcoal. If you now toss a piece of wood, or even a sheet of paper, onto this fire, what you will notice is that the new fuel produces a lot of smoke as it heats up. Then, all of a sudden (often with a small pop), it bursts into flame and the smoke disappears. If you have a fireplace or wood stove, or if you have been around a lot of campfires, this little scene is very familiar to you. It tells you a lot about smoke -- let's look at what is happening.
There are four things that you find in any piece of wood:

- WATER - Freshly cut wood contains a lot of water (sometimes more than half of its weight is water). Seasoned wood (wood that has been allowed to sit for a year or two) or kiln-dried wood contains a lot less water, but it still contains some.
- VOLATILE ORGANIC COMPOUNDS - When the tree was alive, it contained sap and a wide variety of volatile hydrocarbons in its cells. Cellulose (a chief component of wood) is a carbohydrate, meaning it is made of glucose. A compound is "volatile" if it evaporates when heated. These compounds are all combustible (gasoline and alcohol are, after all, hydrocarbons -- the volatile hydrocarbons in wood burn the same way).
- CARBON - the amount of carbon in dry wood will vary depending on the species of wood, age of wood etc. It can be expected that 1 Kg of wood contains about 450 to 500 gm of Carbon.
- ASH - Ash is the non-burnable minerals in the tree's cells, like calcium, potassium and magnesium.
What Type of Fire Extinguisher do I need?
The Fire Extinguisher must be appropriate for the type of fire being fought. Multipurpose fire extinguishers, labeled ABC, may be used on all three classes of fire. If you use the wrong type of extinguisher, you can endanger yourself and make the fire worse. It is also very dangerous to use water or an extinguisher labeled only for Class A fires on a cooking-grease or electrical fire.
Depending on their intended use, portable extinguishers store specific "extinguishing agents," which are expelled onto the fire. The types of Fire Extinguishers include:

- PRESSURIZED WATER MODELS are appropriate for use on Class A fires only. These must never be used on electrical or flammable-liquid fires.
- CARBON DIOXIDE extinguishers contain pressurized liquid carbon dioxide, which turns to a gas when expelled. These models are rated for use on Class B and C fires, but can be used on a Class A fire. Carbon dioxide does not leave a residue.
- DRY CHEMICAL extinguishers are either stored-pressure models or cartridge-operated models. The stored-pressure models have a lever above the handle for operation. The cartridge-operated models require two steps: Depress the cartridge lever, and then squeeze the nozzle at the end of the hose. The dry chemicals leave a residue that must be cleaned up after use.
- AMMONIUM PHOSPHATE dry chemical can be used on Class A, B, and C fires, but should never be used on a fire in a commercial grease fryer because of the possibility of reflash and because it will render the fryer's automatic fire-protection system less effective.
- SODIUM BICARBONATE dry chemical, suitable for fighting Class B and C fires, is preferred over other dry-chemical extinguishers for fighting grease fires. Where provided, always use the extinguishing system first. This also shuts off the heat to the appliance.
- POTASSIUM BICARBONATE, UREA-BASE POTASSIUM BICARBONATE, and POTASSIUM CHLORIDE dry chemical are more effective and use less agent than sodium bicarbonate on the same fire.
- FOAM (or AFFF and FFFP) EXTINGUISHERS coat the surface of a burning flammable liquid with a chemical foam. When using a foam extinguisher, blanket the entire surface of the liquid to exclude the air.
What size Fire Extinguishers are available?

Portable extinguishers are also rated for the size of fire they can handle. This rating is expressed as a number from 1 to 40 for Class A fires and from 1 to 640 for Class B fires. This rating will appear on the label-- 2A:10B:C, for example. The larger the numbers, the larger the fire of a specific class on which the extinguisher can be used (but higher-rated models are often heavier - make sure you can hold and operate an extinguisher before you buy it). No number accompanies an extinguisher's Class C rating. The C on the label indicates only that the extinguisher is safe to use on electrical fires.
Extinguishers for Class D fires must match the type of metal that is burning. These extinguishers do not use numerical ratings. Extinguishers for Class D fires are labeled with a list detailing the metals that match the unit's extinguishing agent.
In Case of Fire, Just Remember the Three A's
- ACTIVATE the building alarm system or notify the fire department by calling 911. Or, have someone else do this for you.
- ASSIST any persons in immediate danger, or those incapable on their own, to exit the building, without risk to yourself.
- Only after these two are completed, should you ATTEMPT to extinguish the fire.
Fire Extinguishers Have Their Limits

Portable extinguishers are not designed to fight large or spreading fires. Even against small fires, they are useful only under certain conditions:
- The operator must know how to use the extinguisher.
- The extinguisher must be within easy reach, in working order, and fully charged.
- The operator must have a clear escape route that will not be blocked by fire.
- The extinguisher must match the type of fire being fought. (Extinguishers containing water are unsuitable for use on grease or electrical fires.)
- The extinguisher must be large enough to put out the fire. Many portable extinguishers discharge completely in as few as eight to ten seconds.
!!! Always be sure the fire department inspects the fire site, even if you think you've extinguished the fire !!!
Only Fight a Fire If ...
- ...the fire is small and contained. The time to use a fire extinguisher is in the early, or incipient, stage of a fire. Once the fire starts to grow or spread, it is best to evacuate the building, closing doors or windows behind you.
- ...you are safe from toxic smoke. If the fire is producing large amounts of thick, black smoke or chemical smoke, it may be best not to try to extinguish the fire. Neither, should you attempt to extinguish the fire in a confined space. Outdoors, approach the fire with the wind at your back. Remember that all fires will produce carbon monoxide and many fires will produce toxic gases that can be fatal, even in small amounts.
- ...you have a means of escape. You should always fight a fire with an exit or other means of escape at your back. If the fire is not quickly extinguished, you need to be able to get out quickly and avoid becoming trapped.
- ...your instincts tell you it's OK. If you do not feel comfortable attempting to extinguish the fire, don't try get out and let the fire department do their job.
Fighting Small Fires on the Job
Fire extinguishers in the workplace should be placed conspicuously and within easy reach so they can be accessed quickly while a fire is still small.
Federal regulations require that employers who provide portable fire extinguishers in the workplace also provide training for their use. Used properly, portable fire extinguishers can save lives and property by putting out a small fire in the workplace or containing one until the fire department arrives.
It is essential that all employees be familiar with the proper use of portable extinguishers and know when and when not to use them. In the event of a fire, employees should respond in accordance with their company's fire-emergency plan. Most employees will evacuate. Certain trained and designated employees will evaluate the fire scene and, if the fire is small and conditions are reasonably safe, use a fire extinguisher to fight the fire. If the fire is large or conditions are unsafe, all employees will evacuate.
Fire Extinguisher Use; Remember P.A.S.S.
- PULL the pin - This unlocks the operating lever and allows you to discharge the extinguisher. Some extinguishers may have other lever-release mechanisms.
- AIM low - Point the extinguisher nozzle (or hose) at the base of the fire.
- SQUEEZE the lever above the handle - This discharges the extinguishing agent. Releasing the lever will stop the discharge. (Some extinguishers have a button instead of a lever.)
- SWEEP from side to side - Moving carefully toward the fire, keep the extinguisher aimed at the base of the fire and sweep back and forth until the flames appear to be out. Watch the fire area. If the fire re-ignites, repeat the process.
NOTE: Always keep your back to an unobstructed exit, stand six to eight feet away from the fire, and follow the PASS (Pull Aim Squeeze Sweep) four-step procedure:

How do multi-class dry chemical fire extinguishers work?
If you ask any firefighter what it takes to create a fire, you will learn that you have to have three things:
- A Fuel - some sort of combustible solid, liquid or gas
- Oxygen to react with the fuel
- Heat- there must be enough heat to get the fuel above its flash point. There is paper on your desk right now that is surrounded by oxygen. It does not burn unless you get the paper hot enough.

If you want to put out a fire, you need to remove one of the three elements. When you watch firefighters battling a forest fire, they generally try to remove fuel or heat. Either they pour water on the fire to reduce the temperature, or they try to bulldoze strips of bare earth to eliminate the fuel.
A carbon dioxide fire extinguisher works by eliminating oxygen and replacing it with carbon dioxide. You could do the same thing with just about any non-oxidizing gas (nitrogen, for example), but carbon dioxide is inexpensive and easy to store.
Another way to cut off oxygen is to throw a blanket over the fire. Covering the fire with dirt or sand does the same thing. You might have heard that you can put out a kitchen fire by throwing baking soda or salt on the fire. Throwing sand would do the same thing, but most people have more salt in their kitchens than they have sand.
Dry chemical fire extinguishers are by far the most common fire extinguishers in the home. They can handle all three types of fires you would find in a kitchen or workshop: combustible solids like wood or paper, combustible liquids like gasoline or grease, and electrical fires. The idea behind a dry chemical fire extinguisher is to blanket the fuel with an inert solid (similar to dirt or sand). A dry chemical extinguisher sprays a very fine power of sodium bicarbonate (normal baking soda), potassium bicarbonate (nearly identical to baking soda), or monoammonium phosphate. These solids coat the fuel and smother the fire.
How Do Photoelectric Smoke Detectors Work?
All smoke detectors consist of two basic parts: a sensor to sense the smoke and a very loud electronic horn to wake people up. Smoke detectors can run off of a 9-volt battery or 120-volt house current.
Occasionally you will walk into a store and a bell will go off as you cross the threshold. If you look, you will often notice that a photo beam detector is being used. Near the door on one side of the store is a light (either a white light and a lens or a low-power laser), and on the other side is a photodetector that can "see" the light.
When you cross the beam of light you block it. The photodetector senses the lack of light and triggers a bell. You can imagine how this same type of sensor could act as a smoke detector. If it ever got smoky enough in the store to block the light beam sufficiently, the bell would go off. But there are two problems here: 1) It's a pretty big smoke detector, and 2) it is not very sensitive. There would have to be a LOT of smoke before the alarm would go off -- the smoke would have to be thick enough to completely block out the light. It takes quite a bit of smoke to do that.
Photoelectric smoke detectors therefore use light in a different way. Inside the smoke detector there is a light and a sensor, but they are positioned at 90-degree angles to one another, like this:
In the normal case, the light from the light source on the left shoots straight across and misses the sensor. When smoke enters the chamber, however, the smoke particles scatter the light and some amount of light hits the sensor:
The sensor then sets off the horn in the smoke detector. Photoelectric detectors are better at sensing smoky fires, such as a smoldering mattress.
How Do Ionization Smoke Detectors Work?

All smoke detectors consist of two basic parts: a sensor to sense the smoke and a very loud electronic horn to wake people up. Smoke detectors can run off of a 9-volt battery or 120-volt house current.
Ionization smoke detectors use an ionization chamber and a source of ionizing radiation to detect smoke. This type of smoke detector is more common because it is inexpensive and better at detecting the smaller amounts of smoke produced by flaming fires.
Inside an ionization detector is a small amount (perhaps 1/5000th of a gram) of americium-241. The radioactive element americium has a half-life of 432 years, and is a good source of alpha particles.
Another way to talk about the amount of americium in the detector is to say that a typical detector contains 0.9 microcurie of americium-241. A curie is a unit of measure for nuclear material. If you are holding a curie of something in your hand, you are holding an amount of material that undergoes 37,000,000,000 nuclear transformations per second. Generally, that means that 37 billion atoms in the sample are decaying and emitting a particle of nuclear radiation (such as an alpha particle) per second. One gram of of the element radium generates approximately 1 curie of activity (Marie Curie, the woman after whom the curie is named, did much of her research using radium).
An ionization chamber is very simple. It consists of two plates with a voltage across them, along with a radioactive source of ionizing radiation, like this:
The alpha particles generated by the americium have the following property: They ionize the oxygen and nitrogen atoms of the air in the chamber. To "ionize" means to "knock an electron off of." When you knock an electron off of an atom, you end up with a free electron (with a negative charge) and an atom missing one electron (with a positive charge). The negative electron is attracted to the plate with a positive voltage, and the positive atom is attracted to the plate with a negative voltage (opposites attract, just like with magnets). The electronics in the smoke detector sense the small amount of electrical current that these electrons and ions moving toward the plates represent.
When smoke enters the ionization chamber, it disrupts this current -- the smoke particles attach to the ions and neutralize them. The smoke detector senses the drop in current between the plates and sets off the horn.
Speaking of alarms, whenever the words "nuclear radiation" are used an alarm goes off in many people's minds. The amount of radiation in a smoke detector is extremely small. It is also predominantly alpha radiation. Alpha radiation cannot penetrate a sheet of paper, and it is blocked by several centimeters of air. The americium in the smoke detector could only pose a danger if you were to inhale it. Therefore, you do not want to be playing with the americium in a smoke detector, poking at it, or disturbing it in any way, because you don't want it to become airborne.


